 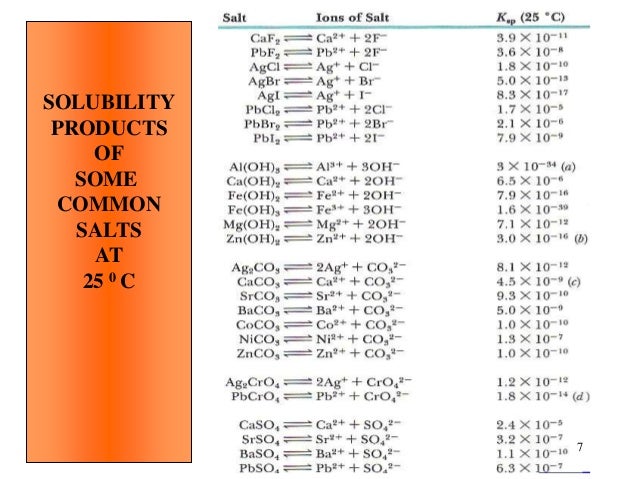
Chromite is the most important chromium ore mined for the recovery of chromium. The most stable oxidation state of the element chromium is chromium (III), found in chromite. 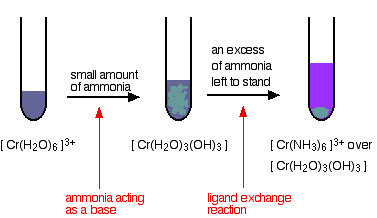
All chromium compounds have no taste or odour. Chromium chloride is a hygroscopic compound with melting point of 1152☌ and specific gravity of 2.8. Chromium acetate is a grey-green to blue green pasty mass. Chromic oxide is a bright green hygroscopic powder with melting point of 2435☌, boiling point of 4000☌ and specific gravity of 5.2. Properties vary widely depending on the particular compound. Synonyms: Chromic ion, Chromium ion, Trivalent chromiumĬhromium sulfate (CASR# 1) Physical propertiesĬhromium is a white, hard, lustrous and brittle metal that is extremely resistant to ordinary corrosive agents. Chromic sulfate is used in tanning, green paints, inks and text dyes and in ceramics. Chromium (III) nitrate is used in the preparation of chrome catalysts, in textile printing operations, and as a corrosion inhibitor. Chromium (III) acetate is used to fix certain textile dyes, to harden photographic emulsions and as a catalyst. More specifically, chromium (III) oxide is used as a paint pigment, a fixative for certain textile dyes and as a catalyst. Chromium (III) compounds are employed as pigment, as catalyst, as tanning agent in the tanning industry, in the production of pure chromium metal and chromium (VI) compounds, and in the production of refractory bricks. More applications are in the photographic industry (sensitiser), in industrial water treatment (including treatment of cooling tower water), as medicinal astringents and antiseptics, and in nuclear and high temperature research. protective coatings for automotive and equipment accessories), as dyes, as inorganic paint pigments, for leather tanning, as fungicides and wood preservatives, and as catalysts. Chromium compounds are used for chrome plating (e.g. The successful synthesis of perovskite with high-Cr content and its structural characterization are of key importance because the study of its thermodynamic constants combined with the data on phase relations in the lower-mantle systems can provide new constraints on thermobarometry of perovskite-bearing assemblages.Chromium metal is used as an alloying element to harden steel and to manufacture stainless steel. Although high Cr-contents are not considered in the pyrolite model, Cr-bearing perovskite may be an important host for chromium in the lower mantle. Such substitutions cause a shortening of the and a lengthening of the distances with respect to the values typically observed for pure MgSiO 3 perovskite. Chromium was found to substitute for both Mg at the dodecahedral X site (with a mean bond distance of 2.187 Å) and Si at the octahedral Y site (mean: 1.814 Å), according to the reaction Mg 2+ + Si 4+ = 2Cr 3+. The structure was refined to R = 0.046 using 473 independent reflections. The crystal structure and chemical composition of a crystal of (Mg 1- xCr x)(Si 1- xCr x)O 3 perovskite (with x = 0.07) synthesized in the model system Mg 3Cr 2Si 3O 12-Mg 4Si 4O 12 at 23 GPa and 1600 ☌ have been investigated. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
